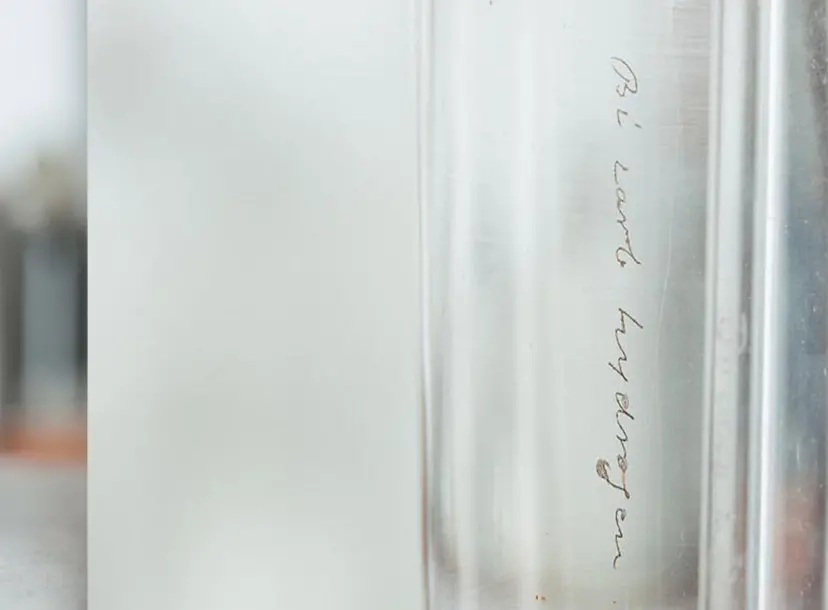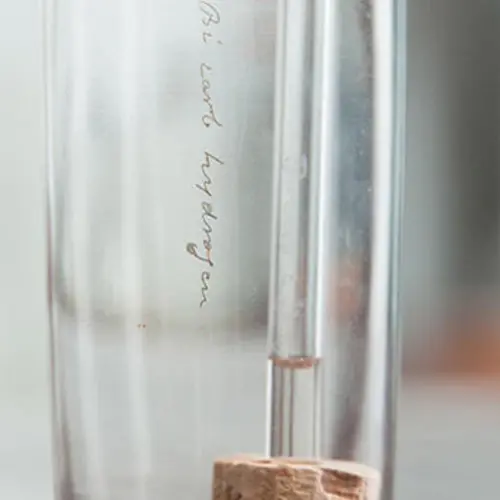Date: 1825
Place made: Basement laboratory at the Ri
Alternative name: Bicarburet of hydrogen
Materials: Glass, Cork, Benzene
Measurements: Height: 146mm, diameter: 44mm
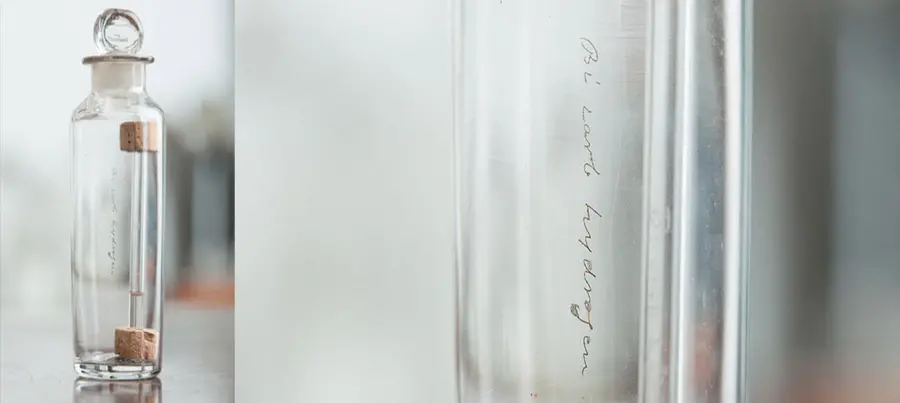
Description
A small vial containing Faraday’s sample of benzene, a colourless liquid with a sweet smell. If you look closely you can see ‘Bicarburet of hydrogen’ scratched onto both the vial and the bottle.
Benzene is a natural hydrocarbon and a component of crude oil. Faraday isolated this substance for the first time in 1825 while investigating an oily residue that was created as a by-product of the production of ‘portable gas’. Portable gas was created by dropping whale or fish oil into a hot furnace, compressing the gas which was created and then storing it in containers to use in lamps in private and public buildings.
Faraday was interested in the liquid which formed when the gas was pressurised. He experimented with distilling this mixture and, as he reported in a paper to the Royal Society on 16th June, he ‘succeeded in separating a new compound of carbon and hydrogen, which I may by anticipation distinguish as bi-carburet of hydrogen’.
By the mid 19th Century benzene was being manufactured on an industrial scale from coal tar. It was mainly used as a solvent for degreasing engines, but by the 20th century, it was in use for everything from aftershave lotions to part of the process of making decaffeinated coffee. The substance is now known to be toxic and is now used mainly in industrial processes to create other substances.
The structure of benzene has been the subject of much research. Many theories were put forward throughout the 19th century but it wasn’t until 1925, a century after its discovery that Kathleen Lonsdale confirmed the structure through x-ray crystallography, using photos taken at the Ri (although not from this sample).
Where can I view this?
This object is currently on display on the lower ground floor of the Royal Institution in the Faraday Museum.